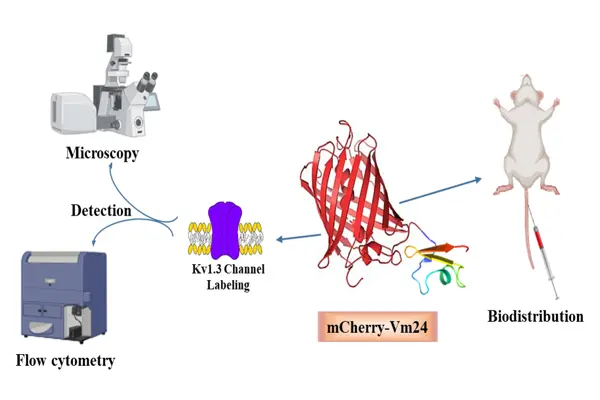
 Changes in the activity of the voltage-gated potassium channel Kv1.3 have been linked to several autoimmune and neurological disorders. To help detect and study cells that produce this channel, we designed a fluorescent molecular probe that binds to Kv1.3 and can be visualized under a microscope. The probe was created by genetically linking two components: Vm24, a small peptide originally isolated from scorpion venom that selectively blocks Kv1.3; and mCherry, a red fluorescent protein commonly used as a biological marker due to its brightness and stability. The resulting fusion protein, mCherry-Vm24, was produced in yeast cells using the Pichia pastoris expression system and purified for further testing. Biochemical and functional analyses confirmed that the fusion protein was correctly formed and biologically active. Although its binding strength to Kv1.3 was weaker than that of the original Vm24 peptide, mCherry-Vm24 still bound very tightly (in the picomolar range). It remained highly selective for Kv1.3 over other related channels and showed nearly irreversible binding. In cell-based experiments, mCherry-Vm24 specifically labeled cells engineered to express human Kv1.3 channels, as demonstrated by confocal microscopy and flow cytometry. In mice, the fusion protein exhibited the strongest fluorescence signal in the kidneys and higher accumulation in the spleen than mCherry alone. Overall, mCherry-Vm24 is a useful tool for detecting and visualizing Kv1.3 channels in cultured cells and living organisms, which supports further research into diseases associated with altered Kv1.3 expression.
Changes in the activity of the voltage-gated potassium channel Kv1.3 have been linked to several autoimmune and neurological disorders. To help detect and study cells that produce this channel, we designed a fluorescent molecular probe that binds to Kv1.3 and can be visualized under a microscope. The probe was created by genetically linking two components: Vm24, a small peptide originally isolated from scorpion venom that selectively blocks Kv1.3; and mCherry, a red fluorescent protein commonly used as a biological marker due to its brightness and stability. The resulting fusion protein, mCherry-Vm24, was produced in yeast cells using the Pichia pastoris expression system and purified for further testing. Biochemical and functional analyses confirmed that the fusion protein was correctly formed and biologically active. Although its binding strength to Kv1.3 was weaker than that of the original Vm24 peptide, mCherry-Vm24 still bound very tightly (in the picomolar range). It remained highly selective for Kv1.3 over other related channels and showed nearly irreversible binding. In cell-based experiments, mCherry-Vm24 specifically labeled cells engineered to express human Kv1.3 channels, as demonstrated by confocal microscopy and flow cytometry. In mice, the fusion protein exhibited the strongest fluorescence signal in the kidneys and higher accumulation in the spleen than mCherry alone. Overall, mCherry-Vm24 is a useful tool for detecting and visualizing Kv1.3 channels in cultured cells and living organisms, which supports further research into diseases associated with altered Kv1.3 expression.
A Fluorescent Protein-Tagged Scorpion Toxin with Picomolar Affinity for the Kv1.3 K+ Channel
Changes in the activity of the voltage-gated potassium channel Kv1.3 have been linked to several autoimmune and neurological disorders.
Last update:
2026. 03. 26. 15:52