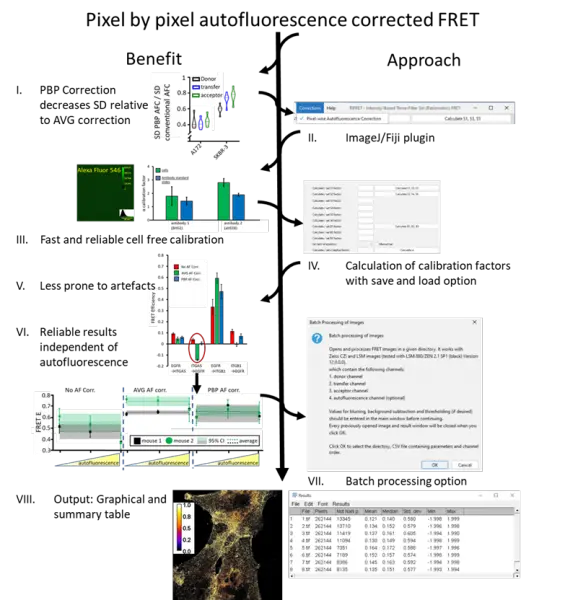
The actual interaction between signaling species in cellular processes is often more important than their expression levels. Förster resonance energy transfer (FRET) is a popular tool for studying molecular interactions, since it is highly sensitive to proximity in the range of 2-10 nm. Spectral spillover-corrected quantitative (3-cube) FRET is a cost effective and versatile approach, which can be applied in flow cytometry and various modalities of fluorescence microscopy, but may be hampered by varying levels of autofluorescence. Here, we have implemented pixel-by-pixel autofluorescence correction in microscopy FRET measurements, exploiting cell-free calibration standards void of autofluorescence that allow the correct determination of all spectral spillover factors. We also present an ImageJ / Fiji plugin for interactive analysis of single images as well as automatic creation of quantitative FRET efficiency maps from large image sets. For validation, we used bead and cell based FRET models covering a range of signal to autofluorescence ratios and FRET efficiencies and compared the approach with conventional average autofluorescene / background correction. Pixel-by-pixel autofluorescence correction proved to be superior in the accuracy of results, particularly for samples with spatially varying autofluorescence and low fluorescence to autofluorescene ratios. This renders the method highly recommended for the case for physiological expression levels and for histopathological diagnosis.
István Rebenku, Cameron B Lloyd, János Szöllősi, György Vereb